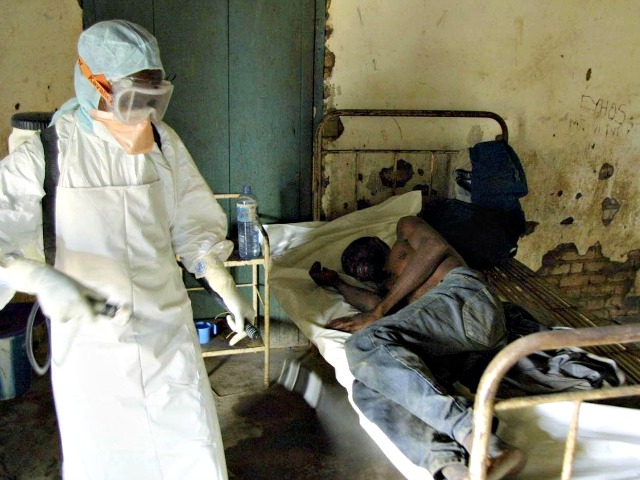
Despite not having been passed through rigorous clinical trials, or in some cases not having been tested on humans at all, three experimental Ebola treatments are going to be deployed in West Africa in coming weeks as part of an enormous international effort to halt the disease that has now killed over 5,000.
Because the situation in West Africa is now becoming so desperate, when the treatments are administered by the three teams they won’t even follow ordinary clinical trial procedures like observing ‘test groups’ who are given placebos. Those treated will instead be compared to the mortality rate of the broader infected population for signs of progress.
One of the trials, funded by British science charity the Wellcome trust will be conducted by a team of scientists from the University of Oxford and will deploy a new American antiviral, brincidofovir, to 150 Ebola patients. The Guardian reports that while the drug has been shown to work against the Ebola virus in vitro, or in isolated laboratory conditions, it hasn’t yet been tested on animals or humans and side effects are unknown.
Speaking about the risks of treating patients with untested drugs, Professor Peter Horby from Oxford’s tropical diseases centre said: “Conducting clinical trials of investigational drugs in the midst of a humanitarian crisis is a new experience for all of us, but we are determined not to fail the people of west Africa. It has been a privilege to witness the extraordinary willingness of all the partners in this initiative to step outside their comfort zones in order to fast-track these critically important trials”.
Two further trials will take place in the former French colony of Guinea. Another antiviral, this time developed by a Japanese company will be trialled the French National Institute of Health and Medical Research. The drug, favipiravir, has been tested on mice in a German laboratory and led to a 100 percent recovery rate of those treated, though again it has not yet been tested on humans. A final test won’t involve any new drugs, but instead the transfer of blood and plasma from Ebola survivors to newly infected carriers, in the hope the introduction of antibodies will help the body fight on its own.
All of the tests are being supported by international medical group Médecins sans Frontières (Doctors Without Borders, MSF) and the World Health Organisation. Dr Annick Antierens of MSF said: “This is an unprecedented international partnership which represents hope for patients to finally get a real treatment against a disease that today kills between 50 and 80 percent of those infected.
“As one of the principal providers of medical care to Ebola patients in West Africa, MSF is taking part in these accelerated clinical trials to give people affected by the current outbreak a better chance of survival”.
The World Health Organisation released it’s latest death and infection figures for the outbreak yesterday, noting that while the present number of known deaths from the virus had exceeded 5,100, the actual number may be far higher. As the eventual mortality rate of Ebola is in excess of 70 percent, that there are now more than 14,000 known cases of infection suggests there is more bad news to come.
COMMENTS
Please let us know if you're having issues with commenting.