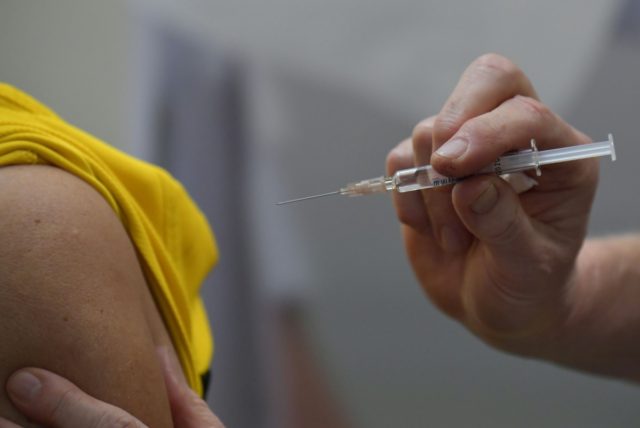
U.S. researchers on Monday reportedly administered the first shot to the first participant in a clinical trial for an experimental vaccine to protect against the deadly novel coronavirus as cases of the disease continue to surge across the globe.
“Going from not even knowing that this virus was out there … to have any vaccine” in testing in about two months is unprecedented, Dr. Lisa Jackson, the leader of the Kaiser Permanente coronavirus vaccine study, told the Associated Press (AP).
“We don’t know whether this vaccine will induce an immune response, or whether it will be safe. That’s why we’re doing a trial,” Jackson stressed. “It’s not at the stage where it would be possible or prudent to give it to the general population.”
On Monday, U.S. President Donald Trump acknowledged that the “first phase” of the trials had begun.
“This is one of the fastest vaccine development launches in history, not even close,” Trump said.
The first vaccine trial marked only the beginning of the process to determine whether the shots are safe and ready for widespread use, which could take up to a year and a half.
AP learned from an unnamed U.S. government official that the coronavirus vaccine trials would start on Monday.
The news outlet revealed:
With a careful jab in a healthy volunteer’s arm, scientists at the Kaiser Permanente Washington Research Institute in Seattle [on Monday] begin an anxiously awaited first-stage study of a potential [coronavirus or] COVID-19 vaccine developed in record time after the new virus exploded from China and fanned across the globe.
U.S. researchers will give the first round of shots to 45 carefully chosen healthy volunteers between the ages of 18 and 55.
AP explained:
Testing will begin with 45 young, healthy volunteers with different doses of shots. … There’s no chance participants could get infected from the shots, because they don’t contain the virus itself. The goal is purely to check that the vaccines show no worrisome side effects, setting the stage for larger tests.
Dr. Anthony Fauci, the director of the National Institute of Allergy and Infectious Diseases (NIAID) has warned that even if initial testing goes well, “you’re talking about a year to a year and a half” before any vaccine could be ready for widespread use.
NIAID is a component of the U.S. National Institutes of Health (NIH).
Finding a vaccine “is an urgent public health priority,” Dr. Fauci declared in a statement Monday.
Fauci noted that the new vaccine study, “launched in record speed, is an important first step toward achieving that goal.”
The NIH and Massachusetts-based biotechnology company Moderna Inc. developed the new vaccine, which is not the only one in the works.
Dozens of Researchers across the world are reportedly racing to develop a vaccine as coronavirus cases continue to increase.
AP noted:
Also in the works: Inovio Pharmaceuticals aims to begin safety tests of its vaccine candidate next month in a few dozen volunteers at the University of Pennsylvania and a testing center in Kansas City, Missouri, followed by a similar study in China and South Korea.
Researchers would reportedly have to try the vaccine on thousands of people fo find out if it genuinely protects against the virus, and it does no harm.
President Trump has been pushing for the rapid development of a vaccine, revealing in recent days that the research is “moving along very quickly.”
Trump hopes that researchers will create a vaccine “relatively soon.”
Currently, there are no proven treatments against the novel coronavirus.

COMMENTS
Please let us know if you're having issues with commenting.