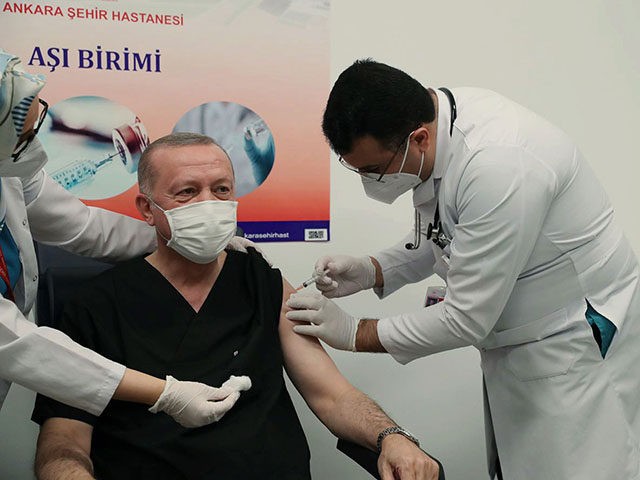
Turkish President Recep Tayyip Erdoğan got his first dose of the Chinese coronavirus vaccine candidate, “CoronaVac,” on Thursday.
Erdoğan received the inoculation at Ankara City Hospital in the national capital, where the procedure was broadcast live on state television. After medical staff administered the president’s shot, Erdoğan told reporters that he believes “all political leaders” should “encourage” the public to receive a Chinese coronavirus vaccine.
The president added that “members of his ruling Justice and Development (AK) Party’s Central Decision and Executive Board were also vaccinated along with him,” according to Turkey’s state-run Anadolu Agency.
Turkey’s official drug and medical equipment body approved CoronaVac for emergency use against the Chinese coronavirus in Turkey on Wednesday. Ankara contracted with CoronaVac’s Chinese maker, SinoVac Biotech, to secure 3 million doses of the vaccine candidate for the Turkish population. The first batch of the 3-million-dose order arrived in Turkey on December 30.
“I hope another 30 million is coming after this three million [doses of the vaccine]. We will reach 50 million doses with those coming from China and so we will continue our fight as we meet this need rapidly,” President Erdoğan said after receiving his first CoronaVac dose on Thursday.
Turkish health minister Fahrettin Koca became the first person in Turkey to receive CoronaVac on Wednesday, followed shortly after by committee members of Turkey’s Coronavirus Scientific Advisory Board. More than 250,000 healthcare workers across Turkey have been vaccinated against the Chinese coronavirus so far, according to Anadolu Agency on Thursday. An estimated 1.1 million health workers across Turkey will be vaccinated against the Chinese coronavirus within a month, Turkey’s health ministry predicted on January 13.
Like Erdoğan, Indonesian President Joko Widodo also received his first dose of CoronaVac on live TV this week. Turkey, Indonesia, Brazil, and other countries have contracted with China’s Sinovac Biotech to use its coronavirus vaccine candidate as part of national vaccination drives. Reports of CoronaVac’s efficacy rating have varied significantly, causing scientific experts to question its viability.
Late-stage clinical trials in Indonesia found CoronaVac to be 65.3 percent effective against the Chinese coronavirus on January 12, while Brazilian scientists partnered with Sinovac said on the same day that final data showed CoronaVac had an efficacy rate of 50.38 percent. The same Brazilian researchers had previously claimed on January 7 that CoronaVac was 78 percent effective against the virus.
CoronaVac works by using killed viral particles to expose the body’s immune system to the Chinese coronavirus. It requires two separate doses spaced 28 days apart.

COMMENTS
Please let us know if you're having issues with commenting.