Beijing’s government announced on Wednesday plans to enforce a Chinese coronavirus vaccine mandate on select members of its population of roughly 21.3 million starting next week, China’s state-run Global Times reported.
“To further implement normalized epidemic prevention and control, consolidate achievements and protect the health and safety of the public, especially the elderly and public service workers, Beijing plans to roll out a series of epidemic prevention and control measures,” the newspaper reported on July 6.
“The new regulations stipulate that from July 11, 2022, Beijing residents should be vaccinated (with exceptions for those not suited to vaccination) before entering public venues, including in-person training institutions, libraries, museums, cinemas, art galleries, culture centers, sports venues, gyms, performance and entertainment settings and internet cafes,” the Global Times relayed.
“Besides, elderly people who enter colleges and venues offering entertainment should also be vaccinated if they are eligible,” the Chinese Communist Party-controlled newspaper added.
Party officials claim that Beijing has been battling an outbreak of the Chinese coronavirus in recent weeks. The Chinese national capital’s epidemic is part of a nationwide resurgence of the disease that began this year. In response, China’s ruling Communist Party, which obeys a strict “zero tolerance” policy toward the disease, has imposed snap lockdowns on some of its largest communities.
The policy forced Shanghai’s government to confine all of its 25 million-plus residents to their homes for 65 consecutive days after new Chinese coronavirus infections emerged in the city in mid-March. While Shanghai’s municipal government claimed that the city’s stay-at-home order lasted from March 28 to June 1, movement restrictions remained in place across the financial hub throughout June, crippling its normally lucrative economy. Shanghai announced a new, snap lockdown across 12 of the city’s 16 residential districts on July 5, so that municipal health workers could conduct mass testing for the Chinese coronavirus amid a fresh outbreak of the disease.
The Global Times on July 6 did not specify what type of Chinese coronavirus vaccines Beijing would require residents to receive as part of its looming vaccine mandate.
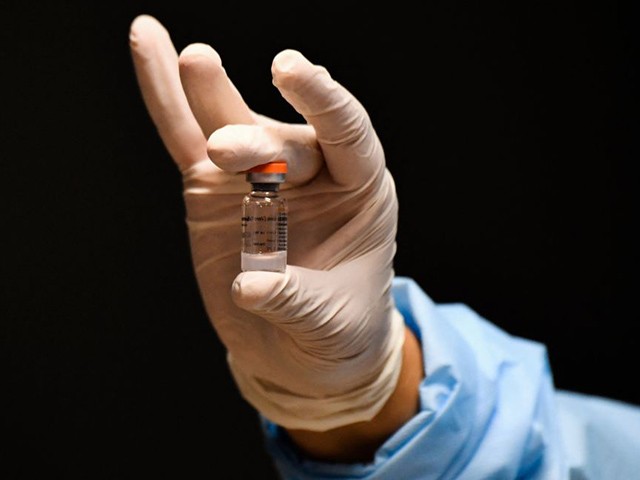
A nurse holds a vial of the CoronaVac vaccine -developed by China’s Sinovac laboratory- against the COVID-19 disease, in Bogota on March 9, 2021. (Photo by JUAN BARRETO/AFP via Getty Images)
China’s health ministry has approved six different types of domestically developed Chinese coronavirus vaccines and vaccine candidates for use in the country. These include offerings by the following Chinese or Chinese state-owned pharmaceutical and biotech companies: Anhui Zhifei Loongcom (“Zifivax,” based on protein subunit technology); CanSino (“Convidecia,” based on non-replicating viral vector technology); Shenzhen Kangtai Biological Products Co. (“KCONVAC,” based on inactivated vaccine technology); Sinopharm Beijing (“Covilo,” based on inactivated vaccine technology); Sinopharm Wuhan (“Inactivated Vero Cells,” based on inactivated vaccine technology); and Sinovac (“CoronaVac,” based on inactivated vaccine technology).
The World Health Organization (W.H.O.) explains inactivated vaccine technology on its website, writing:
The first way to make a vaccine is to take the disease-carrying virus or bacterium, or one very similar to it, and inactivate or kill it using chemicals, heat or radiation. This approach uses technology that’s been proven to work in people – this is the way the flu and polio vaccines are made – and vaccines can be manufactured on a reasonable scale.
However, it requires special laboratory facilities to grow the virus or bacterium safely, can have a relatively long production time, and will likely require two or three doses to be administered.
A protein subunit vaccine “only uses the very specific parts (the subunits) of a virus or bacterium that the immune system needs to recognize,” according to the W.H.O. “It doesn’t contain the whole microbe or use a safe virus as a vector. The subunits may be proteins or sugars. Most of the vaccines on the childhood schedule are subunit vaccines, protecting people from diseases such as whooping cough, tetanus, diphtheria and meningococcal meningitis.”
The U.N. international public health body explains viral vector vaccine technology as using “a safe virus to deliver specific sub-parts – called proteins – of the germ of interest so that it can trigger an immune response without causing disease.”
“To do this, the instructions for making particular parts of the pathogen of interest are inserted into a safe virus. The safe virus then serves as a platform or vector to deliver the protein into the body. The protein triggers the immune response. The Ebola vaccine is a viral vector vaccine and this type can be developed rapidly,” according to the W.H.O.
The W.H.O. criticized China’s “zero tolerance” policy toward the Chinese coronavirus and its associated lockdowns as “unsustainable” on May 10.

COMMENTS
Please let us know if you're having issues with commenting.