President Joe Biden’s Department of Justice is asking the Supreme Court to reverse a lower court decision halting two U.S. Food and Drug Administration (FDA) actions that loosened restrictions around mifepristone, the first pill used in a two-drug chemical abortion regimen.
Solicitor General Elizabeth Prelogar told the high court in a petition for a writ of certiorari filed on Friday that if the lower court decision is allowed to take effect, it would “up-end the regulatory regime for mifepristone with damaging consequences for women seeking lawful abortions and a healthcare system that relies on the availability of the drug under the current conditions of use.”
“And the logic of the Fifth Circuit’s unprecedented decision would threaten to severely disrupt the pharmaceutical industry and prevent FDA from fulfilling its statutory responsibilities according to its scientific judgment,” the petition reads.
Danco Laboratories LLC, which distributes mifepristone under the brand name Mifeprex, also filed a cert petition on Friday, the company announced in a press release.
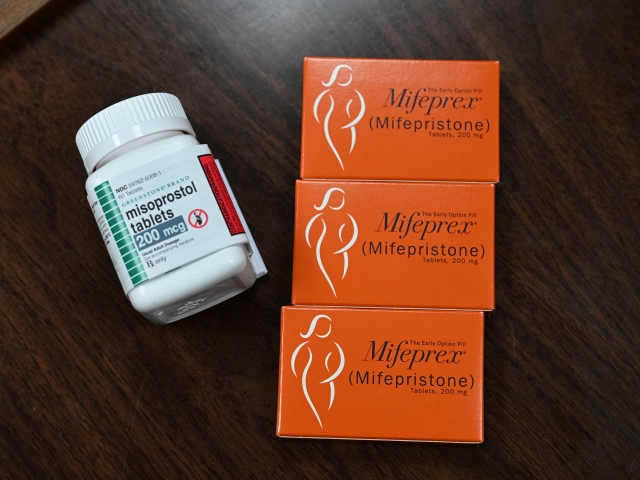
Mifepristone (Mifeprex) and Misoprostol, the two drugs used in a medication abortion, are seen at the Women’s Reproductive Clinic. (ROBYN BECK/AFP via Getty Images)
“As Danco’s petition explains, the case presents a serious question: whether courts can disregard constitutional and statutory limits on judicial review of agency action to overrule agency decisions that they dislike,” the company said in a statement. “Danco asks the Court to grant review of both the determination that doctors who do not prescribe or want to prescribe Mifeprex® have standing and the determination that FDA acted unreasonably in approving the changes in 2016 and 2021 despite the extensive study and other data supporting those decisions.”
The petitions come after a three-judge panel of the conservative U.S. Court of Appeals for the Fifth Circuit ruled in August that the FDA’s 2016 decision to allow the abortion pill to be taken later in pregnancy is unlawful. The court said the same of the FDA’s 2021 rule change, which allowed the abortion pill to be mailed directly to patients and allowed medical professionals other than doctors to prescribe mifepristone.
“In loosening mifepristone’s safety restrictions, FDA failed to address several important concerns about whether the drug would be safe for the women who use it,” reads the panel opinion, penned by Judge Jennifer Walker Elrod.
“It failed to consider the cumulative effect of removing several important safeguards at the same time. It failed to consider whether those ‘major’ and ‘interrelated’ changes might alter the risk profile, such that the agency should continue to mandate reporting of non-fatal adverse events,” the opinion continued. “And it failed to gather evidence that affirmatively showed that mifepristone could be used safely without being prescribed and dispensed in person.”
Elrod wrote that at the preliminary stage, plaintiffs “have made a substantial showing” that the 2016 and 2021 rule changes violate the Administrative Procedure Act (APA).
Despite the Fifth Circuit’s ruling against the government and the drug manufacturer, mifepristone has remained available under existing regulations while the litigation continues. The Supreme Court preemptively paused any ruling from an appeals court this spring, pending a petition for the Supreme Court to take the case. If the Supreme Court does not take the case, the Fifth Circuit’s order will go into effect.
The Alliance Defending Freedom (ADF) filed the lawsuit in November of 2022 against the FDA on behalf of four national medical associations and several doctors, alleging the agency “chose politics over science and approved chemical abortion drugs for use in the United States.”
Related: NJ Gov. Murphy: We’ll Defy SCOTUS “to Save Lives” if They Rule Against Abortion Pill
The lawsuit points to six discrete agency actions since the legalization of mifepristone and misoprostol in 2000. The ADF alleges that the agency was only able to approve the drug by falsely classifying pregnancy as an “illness.” The lawsuit also alleges that the FDA never studied the safety of mifepristone under the labeled conditions of use, ignored the potential impacts of the hormone-blocking regimen on the developing bodies of adolescent girls, disregarded evidence that chemical abortion drugs cause more complications than surgical abortion, and eliminated necessary safeguards for pregnant girls and women who take the regimen.
The lawsuit additionally details how, in 2016, the FDA extended the permissible gestational age of the baby for which a girl or woman may take the abortion drugs — from seven weeks gestation to ten weeks gestation. Then, in 2021, the FDA allowed abortionists to send mifepristone through the mail, which the ADF says was “in direct violation of federal law.” The FDA recently made permanent its rule to allow women and girls to receive a prescription for mifepristone via telemedicine.
The complaint alleges:
All of the FDA’s actions on chemical abortion drugs — the 2000 approval, the 2016 major changes, the 2019 generic drug approval, and the two 2021 actions to eliminate the in-person dispensing requirement — failed to acknowledge and address the federal laws that prohibit the distribution of chemical abortion drugs by postal mail, express company, or common carrier. Instead, the FDA’s actions permitted and sometimes even encouraged these illegal activities.
According to former abortionist Dr. Anthony Levatino, mifepristone blocks the action of progesterone, which the mother’s body produces to nourish the pregnancy. When progesterone is blocked, the lining of the mother’s uterus deteriorates, and blood and nourishment are cut off to the developing baby, who then dies inside the mother’s womb. The drug misoprostol (also called Cytotec) then causes contractions and bleeding to expel the baby from the mother’s uterus.
Pro-abortion activists protest in front of the Supreme Court on June 26, 2022, in Washington, DC. (Nathan Howard/Getty Images)
The pro-abortion Guttmacher Institute found that mifepristone is used for more than half of all abortions in the United States. In 2020, the drug accounted for 53 percent of all abortions, up from 39 percent in 2017.
The case is FDA v. Alliance for Hippocratic Medicine, No. 23-235 in the Supreme Court of the United States.
Katherine Hamilton is a political reporter for Breitbart News. You can follow her on Twitter @thekat_hamilton.


COMMENTS
Please let us know if you're having issues with commenting.